You have successfully logged out.
Not registered yet?
Infusion Therapy
Infusion therapy with B. Braun for improved efficiency
Infusion therapy plays an important role in patient care. B. Braun offers a holistic infusion therapy concept which combines high-quality products with intelligent technologies and services. Our goal is to make even the most complex therapies simpler, smarter and safer.
Medical Professional
This information is meant for medical professionals only. Please confirm that you are a medical professional before accessing the information.
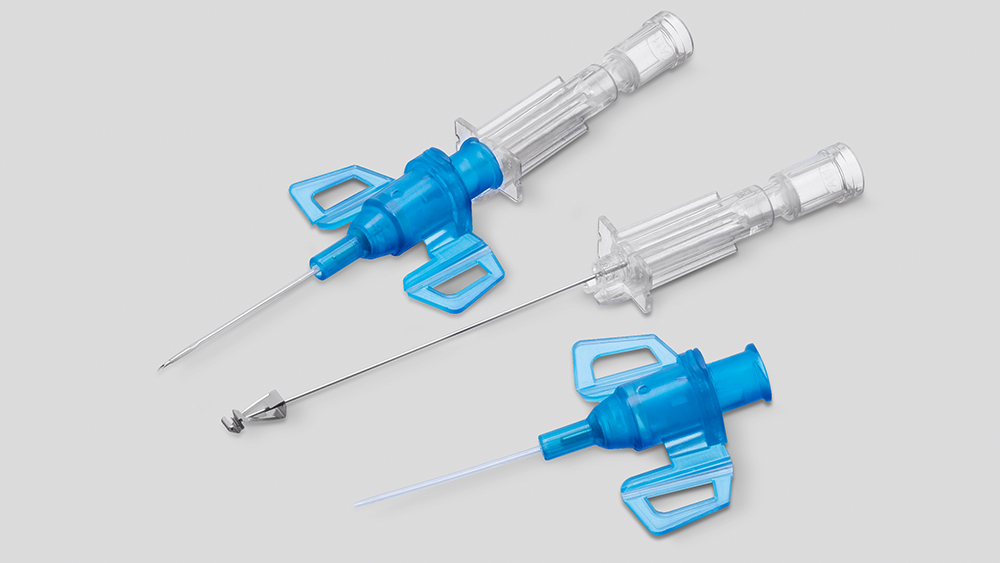
Confirm Yes, I am a health care professional. Cancel No, I am not a health care professional.Closed IV Catheter with Blood Control Septum
Designed to protect against blood exposure and needlestick injuries. Introcan Safety® 3 combines innovative features to protect patients and clinicians.

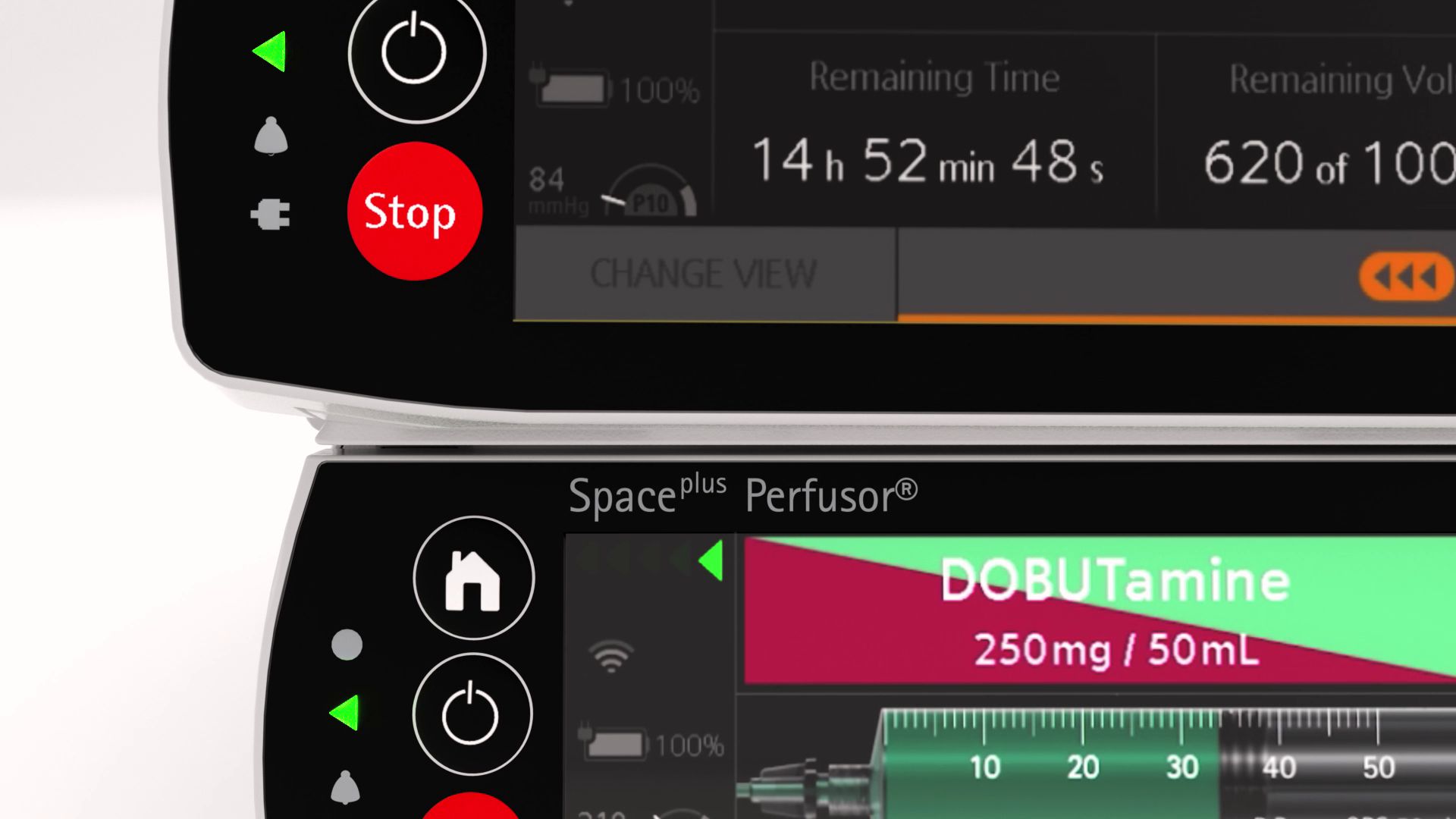
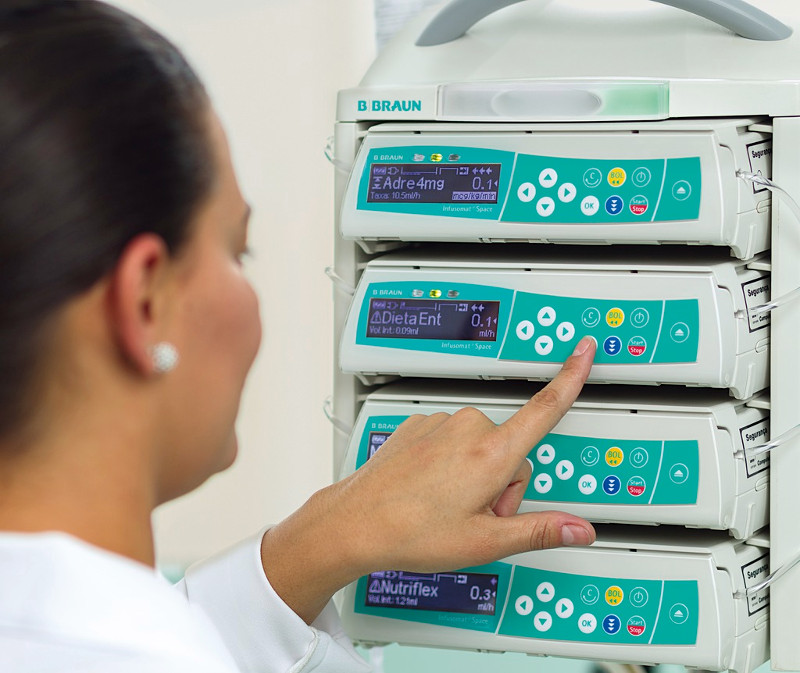
Reducing infusion errors
Reducing chemotherapy mistakes
Errors in providing chemotherapy to patients can have fatal consequences, given the toxicity of the drugs. Know the risks and the preventive strategies for best possible patient care.
Medication errors in chemotherapy
-
> 0k
deaths per year are due to medication error in the US1

Reducing underdosing
Underdosing is an often overlooked and underestimated type of medication error resulting from dead or residual volume. However, there are ways that help to minimise the decreased treatment responses it can cause.
How to reduce underdosing
-
Up to
0%
of antibiotic infusions are underdosed2

Safety first in drug preparation
Hospital pharmacies prepare drugs using manual and automated compounding processes. Which of the two is suitable depends on their advantages and limitations. Risk and productivity investigations are essential.

At eye-level with tiny giants
Young patients certainly belong to one of the most challenging patient groups. Learn more about handling the high risk of error caused by their special anatomy, minimum tolerance ranges, and less patient compliance.
Care for paediatric & neonatal patients
Basic Concepts of Fluid and Electrolyte Therapy
Written by a team of experts in fluid therapy and gastrointestinal surgery, nutrition and renal medicine. The book “Basic Concepts of Fluid and Electrolyte Therapy" provides a practical reference for day-to-day matters related to questions of fluid prescribing and electrolyte management.
Authors: Dileep N. Lobo, Andrew J. P. Lewington, Simon P. Allison
Download for free

Other topics related to Infusion Therapy
Sharing expertise with health care professionals
[1] Makary, M. A., Daniel M. Medical error–the third leading cause of death in the U.S. BMJ 2016; 353.
[2] Rachel Dixon, 2015, Are you under infusing IV antibiotic infusions?, <https://www.england.nhs.uk/signuptosafety/wp-content/uploads/sites/16/2015/11/su2s-infusing-iv-antibiotics-webinar.pdf>, accessed 22.2.22
Infusion Therapy
We will answer your question or feedback as soon as possible.